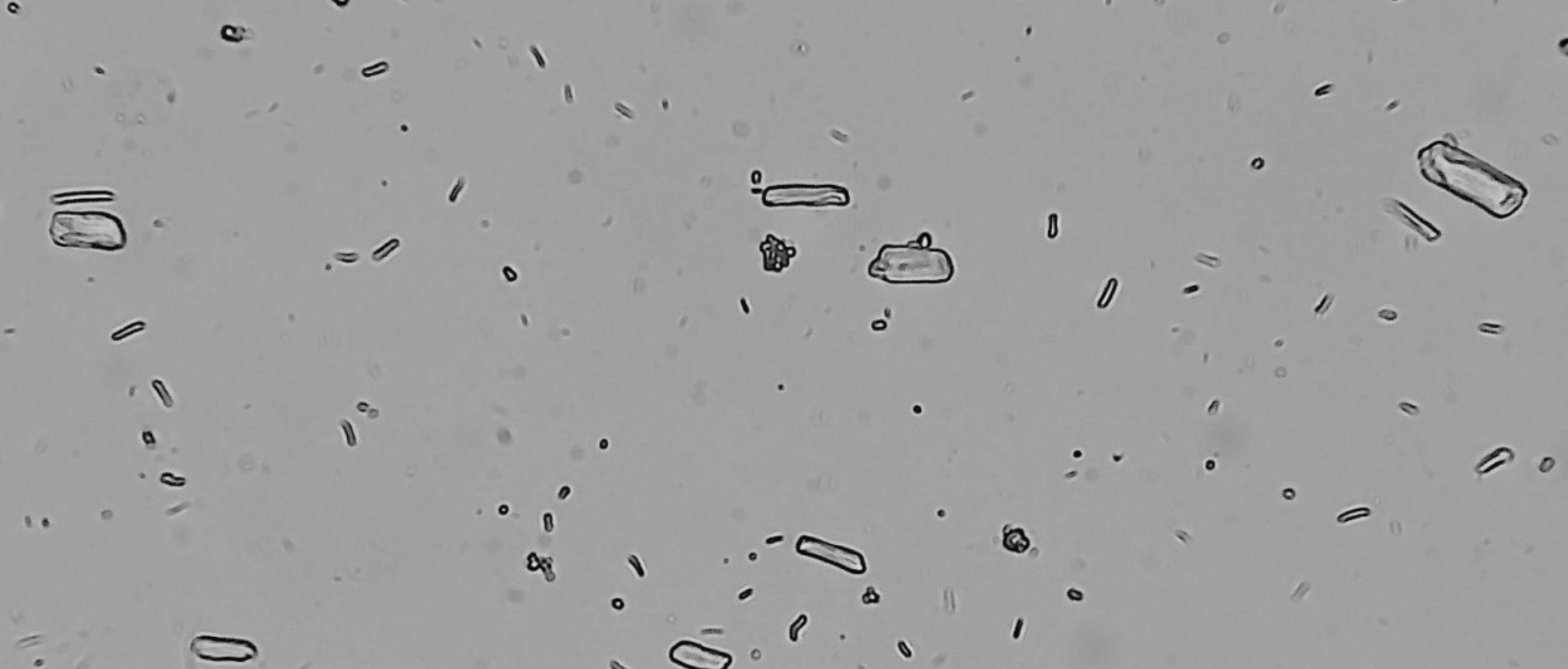
In the pharmaceutical industry, API particle characterization provides a detailed understanding of the size, shape, and structure of a drug compound. These characteristics impact powder flowability, porosity, bulk density, hygroscopicity, blend uniformity, and compressibility—all of which influence key manufacturing steps such as granulation, mixing, compression, and coating.
Crystal size distribution (CSD) or particle size distribution (PSD) plays a crucial role in determining drug solubility, dissolution rate, and shelf life. Accurate particle characterization helps maintain consistent product quality and therapeutic efficacy.